MSKCC DCIS Nomogram Calculator: Expert Guide for Breast Surgeons
An expert clinical guide on using the MSKCC DCIS Nomogram to predict local recurrence risk and guide shared decision-making in breast surgery.
Quick Navigation
- 1. Introduction: Individualized Risk Assessment in DCIS
- 2. What Is the MSKCC DCIS Nomogram?
- 3. Why the MSKCC DCIS Nomogram Matters in Contemporary Care
- 4. Clinical Evidence, Validation, and Genomic Integration
- 5. High-Risk Features and Embedded Reference Data
- 6. Operating the OncoToolkit MSKCC DCIS Calculator
- 7. Leveraging the Platform for Clinical Care, Teaching, and Research
- 8. Clinical FAQ: High-Value Questions for Breast Surgeons
- 9. Start Using the MSKCC DCIS Nomogram on OncoToolkit
1. Introduction: Individualized Risk Assessment in DCIS
Ductal carcinoma in situ (DCIS) now accounts for 20–25% of screen-detected breast lesions in many programmes, yet its biologic heterogeneity makes local therapy decisions uniquely challenging. Ten‑year disease‑specific survival typically exceeds 98%, but rates of ipsilateral breast tumor recurrence (IBTR) after breast-conserving surgery (BCS) vary from under 5% to more than 25% depending on age, grade, necrosis, margin width, and other features. At OncoToolkit, we’ve built an MSKCC DCIS Nomogram calculator to convert this multidimensional profile into an individualized 5–10 year local recurrence risk range for surgery alone, giving breast surgeons a concrete baseline when weighing the incremental value of radiotherapy, endocrine therapy, or additional surgery.1, 2, 3, 4, 5, 6, 7, 8
Clinicians are increasingly expected to engage patients in shared decision‑making about de‑escalation options, including radiation omission and, in trial contexts, active surveillance for carefully selected low‑risk DCIS. Without structured tools, it is difficult to communicate individualized risk in a way that is both numerically coherent and aligned with patient values. OncoToolkit’s MSKCC DCIS Nomogram is designed to reduce cognitive load by providing a mobile‑responsive, visually intuitive interface that mirrors real pathology reports, while also allowing results to be saved and exported into MDT documentation or research registries.9, 2, 3, 7, 10, 11

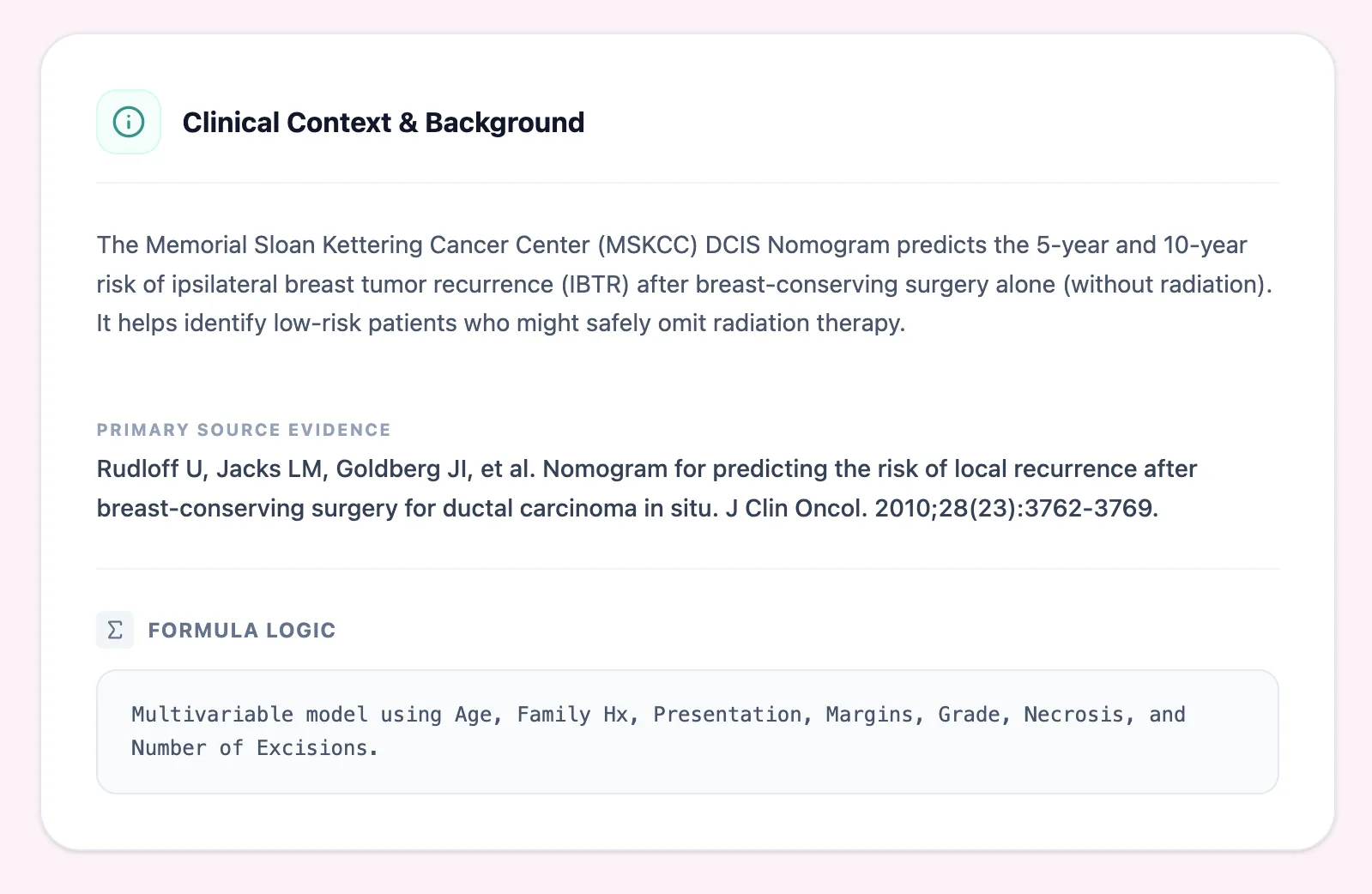
Figure 1. The clinical background panel on OncoToolkit summarises the purpose of the MSKCC DCIS Nomogram and cites the original J Clin Oncol study, helping surgeons quickly recall what endpoint is being predicted and under which treatment assumptions.
2. What Is the MSKCC DCIS Nomogram?
The Memorial Sloan Kettering Cancer Center (MSKCC) DCIS Nomogram is a multivariable prediction model that estimates a woman’s risk of ipsilateral breast tumor recurrence after BCS for DCIS when no adjuvant radiotherapy is given. The endpoint includes both recurrent DCIS and invasive ipsilateral events over 5 and 10 years, giving surgeons a clinically meaningful metric for local control that can be compared directly with trial outcomes.4, 5, 12, 13
The original nomogram incorporates several clinicopathologic variables:
- • Age at diagnosis
- • First‑degree family history of breast cancer
- • Method of detection (screening vs symptomatic)
- • Margin status and margin width
- • Nuclear grade
- • Presence of necrosis
- • Number of excisions required
- • Year of diagnosis and treatment details
OncoToolkit focuses the implementation on the core variables that are universally available and drive most of the prognostic signal:
- • Age at diagnosis (categorical bands)
- • First‑degree family history (yes/no)
- • Detection method (radiologic screening vs symptomatic presentation)
- • Margin status and width, including a specific “Close < 2 mm” category
- • Nuclear grade (low/intermediate vs high)
- • Necrosis (present/absent)
- • Number of excisions to achieve final margins
These inputs are passed through an internal representation of the published Cox model, and the output is deliberately expressed as a percentage range (for example, “10–20% 5/10‑year IBTR risk without radiation”) rather than a single number, acknowledging uncertainty and avoiding overprecision. The software then maps risk to low, intermediate, or high bands displayed on a colour‑coded scale that surgeons can immediately interpret in MDT meetings and patient encounters.8, 4, 7
3. Why the MSKCC DCIS Nomogram Matters in Contemporary Care
3.1 Context: from “treat all” to risk‑adapted de‑escalation
Historically, most women with DCIS underwent surgery plus radiation and often endocrine therapy, reflecting early trials that showed large relative reductions in IBTR with radiotherapy regardless of clinicopathologic risk. However, as longer‑term data emerged, it became clear that absolute benefits vary markedly: women with small, low‑ or intermediate‑grade, widely excised DCIS have low baseline recurrence risk, meaning that even a 50% relative reduction may translate into a modest absolute gain. More recent guidelines and narrative reviews emphasise tailoring local therapy to individual risk and quality‑of‑life considerations, including surgical morbidity, cosmetic outcomes, and the psychological impact of repeated treatments.14, 15, 2, 3, 5, 6, 16, 17, 18, 8
For example:
- The ECOG‑ACRIN E5194 and RTOG 9804 studies showed that carefully selected low‑risk DCIS treated with lumpectomy alone can have 20‑year invasive recurrence rates low enough to make radiation omission reasonable for some women.5, 16, 18
- Pooled cohort analyses involving thousands of patients have quantified how margin width and lesion size jointly influence recurrence risk, suggesting clinically relevant cut points around 2 mm.6, 19, 20
- Ongoing de‑escalation trials and decision‑aid studies are exploring active surveillance strategies and patient preference‑sensitive frameworks, particularly in older or comorbid patients.21, 3, 9
In this landscape, the MSKCC DCIS Nomogram provides a numerical anchor for shared decision‑making: rather than telling a patient she is “low risk,” the surgeon can say, for example, “Your estimated 10‑year chance of the cancer coming back in this breast with surgery alone is in the 5–10% range,” then discuss how radiation or endocrine therapy might modify that baseline.22, 4, 8
3.2 Reducing cognitive load in busy breast clinics
Treatment decisions for DCIS are made in an environment of information overload—radiology, pathology, guidelines, genomic reports, and patient preference data all compete for attention. Workflow analyses show that clinicians frequently rely on heuristics rather than fully processing all available information, especially when digital tools have inconsistent interfaces or are separate from routine documentation systems.23, 24, 11, 25
OncoToolkit intentionally standardises the design language across its calculators, including the MSKCC DCIS Nomogram, Nottingham Prognostic Index, and Oncotype-related tools. Age bands, margin labels, and detection categories are phrased to match real pathology reports (“Close < 2 mm,” “Radiologic [Screening]”), reducing the mental translation needed to use the tool. By allowing results to be saved and exported, the calculator fits naturally into MDT notes and research spreadsheets rather than existing as a disconnected web page, which in turn encourages consistent use and lowers “calculator fatigue.”10, 11, 26, 27, 23, 7
4. Clinical Evidence, Validation, and Genomic Integration
4.1 Model derivation: Cox hazards, not a simple points score
Rudloff et al. used Cox proportional hazards modelling to estimate the effect of each variable on time to ipsilateral breast event, adjusting for competing risks and treatment differences across the cohort. Variables such as high nuclear grade, necrosis, positive/close margins, and younger age carried positive coefficients (increased hazard), while wide negative margins and older age were protective; the model then converted the linear predictor into 5‑ and 10‑year recurrence probabilities via the baseline survival function.13, 4, 12
This approach has several advantages: accounting for the time dimension of recurrence, allowing non‑additive contributions of variables, and being recalibratable as hazards shift.28, 8
4.2 Performance and external validation
Multiple external studies have evaluated how well the MSKCC DCIS Nomogram performs outside the original MSKCC cohort: European validation series showed concordance indices in the 0.63–0.68 range, with generally good calibration; Asian cohorts reported that the nomogram remained predictive, though absolute recurrence rates were influenced by screening uptake differences.17, 29, 30
4.3 Integration with genomic assays and novel decision tools
Several genomic tools—such as the Oncotype DX DCIS Score and DCISionRT—aim to refine IBTR risk estimates. Comparative work shows that genomic scores and the MSKCC nomogram are concordant in many low‑ and high‑risk patients, but discordance arises in intermediate clinicopathologic risk, where genomic tools may reclassify women.32, 33, 34, 26
4.4 Limitations and the importance of judgement
Even with strong validation, the nomogram has clear boundaries: it is designed for pure DCIS treated with BCS; it predicts IBTR, not distant recurrence or overall survival; and it does not directly incorporate HER2 status or hormone receptors.1, 4, 18, 8
5. High-Risk Features and Embedded Reference Data

Figure 2. The reference data tile highlights that age under 40, positive or close margins, high grade or necrosis, and symptomatic presentation are associated with higher recurrence risk, echoing evidence from pooled cohort and margin‑analysis studies.
The embedded reference panel distils decades of observational research into a simple reminder: Young age (< 40 years) is a robust predictor of increased local recurrence; Positive or close margins (< 2 mm) substantially increase risk; and High nuclear grade and necrosis are consistently associated with greater recurrence risk.2, 14, 19, 20, 6
6. Operating the OncoToolkit MSKCC DCIS Calculator
6.1 Input workflow: clinically natural data entry
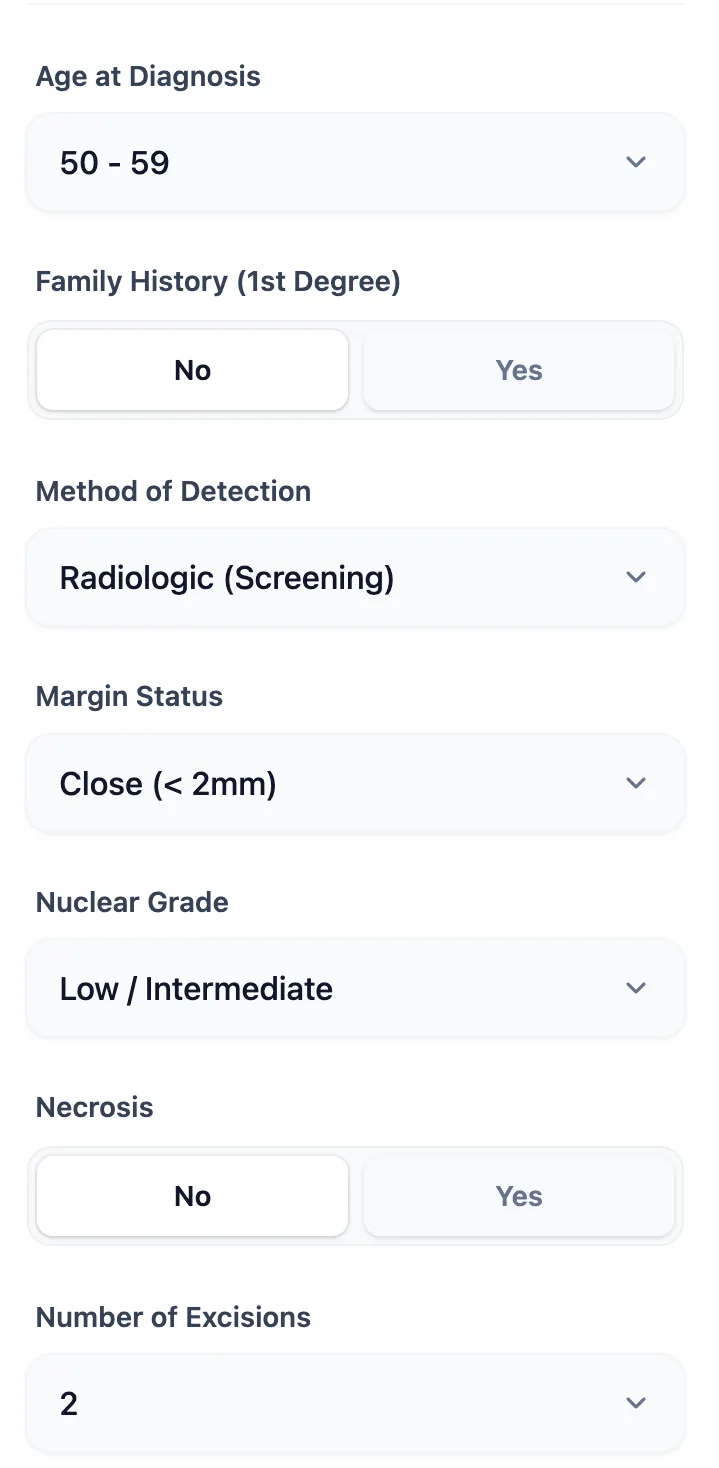
Figure 3. The input form is structured to match real-world pathology and operative notes: age bands, binary family history, detection method, margin categories, grade, necrosis, and number of excisions can all be entered rapidly during clinic or MDT review.
6.2 Output: risk score, range, narrative, and workflow integration

Figure 4. The result view combines a numerical risk score, a colour‑coded low/intermediate/high bar, a 5–10 year local recurrence risk range, and a brief explanation of key risk drivers, with options to save and export the calculation.
7. Leveraging the Platform for Clinical Care, Teaching, and Research
8. Clinical FAQ: High‑Value Questions for Breast Surgeons
Can the MSKCC DCIS Nomogram be used for patients who ultimately receive radiotherapy?
The nomogram predicts IBTR after BCS without radiotherapy. In practice, surgeons apply the relative risk reductions observed in randomized trials—typically around 50%—to the baseline range to approximate risk with radiation.22, 5, 8, 4
How does the MSKCC DCIS Nomogram compare with VNPI and Oncotype/biomarker tools?
The VNPI is a point‑based system; the MSKCC Nomogram yields continuous probabilities. Genomic tools add molecular information; studies suggest high concordance in many patients but relevant discordance in intermediate‑risk cases.26, 34, 32
9. Start Using the MSKCC DCIS Nomogram on OncoToolkit
To integrate individualized DCIS local recurrence risk into your clinical workflow, open the calculator at https://oncotoolkit.com/calculator/mskcc-dcis and bookmark it on your clinic and MDT workstations. Try running it on a spectrum of your recent cases—screen‑detected, low‑grade DCIS with wide margins; symptomatic, high‑grade lesions with necrosis; and multi‑excision borderline scenarios—and compare the predicted 5–10 year IBTR ranges with your current recommendations for radiation, endocrine therapy, or additional surgery.6, 7, 14, 4, 5
References
- Tariq et al. (2021). Clinical outcomes of DCIS. Source
- Japanese Journal of Clinical Oncology (2025). Source
- ScienceDirect (2025). DCIS Systematic Review. Source
- Van Zee et al. (2010). MSKCC Nomogram Derivation. Source
- Wapnir et al. (2015). RTOG 9804 Outcomes. Source
- BMJ (2023). Margin Width and DCIS. Source
- OncoToolkit. MSKCC DCIS Calculator Interface. Source
- Tuttle et al. (2024). De-escalation strategies. Source
- AACR (2022). Decision Aids for DCIS. Source
- OncoToolkit Home. Source
- JAMIA Open (2024). Decision Support Workflow. Source
- Rudloff et al. (2010). J Clin Oncol. Source
- Di Saverio et al. (2012). External Validation. Source
- JAMA Network Open (2023). Source
- Collins et al. (2016). Quality of Life in DCIS. Source
- NPJ Breast Cancer (2024). Low-risk management. Source
- European Cohort Study (2024). Source
- Current Problems in Surgery (2023). Source
- SSO/ASTRO Consensus (2024). Source
- Morrow et al. (2016). Margin Width Meta-analysis. Source
- COMET Trial (2023). Active Surveillance. Source
- JOP (2016). Risk Communication. Source
- Nature Communications (2022). Source
- Nature (2021). Training Surgeons. Source
- Frontiers in Digital Health (2021). Source
- CMAJ (2019). Genomic Assays in DCIS. Source
- Cancers (2022). Prediction Model Review. Source
- Clinical Oncology (2014). European Calibration. Source
- Asian Validation Study (2014). Source
- International Journal of Radiation Oncology (2014). Source
- Scientific Reports (2025). Source
- Cleveland Clinic (2024). Endocrine therapy in DCIS. Source
- Breast Cancer Research (2024). Source
- ASBrS (2019). Genomic vs Nomogram. Source
- Therapeutic Advances in Medical Oncology (2022). Source
- Nature Review Clinical Oncology (2024). Source
- AJR (2007). Symptomatic vs Screened DCIS. Source
- Frontiers in Psychology (2022). Source
- JAMA Network Open (2023). Decisional Conflict. Source
- Trials (2022). Patient decision aids. Source
- Health Expectations (2024). Source
Ready to Simplify Your DCIS Risk Assessment?
Convert complex clinicopathologic variables into an individualized recurrence range in seconds.
Try the MSKCC DCIS Calculator
Free to use. No registration required.