PREDICT Breast (Prognostic Index) Calculator: A Complete Guide for Breast Surgeons
A comprehensive guide for breast surgeons on using the PREDICT model (v4.0) to estimate survival and inform adjuvant therapy decisions.
Quick Navigation
1. Introduction to PREDICT Breast Prognostic Index
Breast cancer treatment planning involves synthesizing multiple tumor and patient variables—age, tumor size, nodal involvement, receptor status, and proliferation markers—into actionable prognostic estimates. For breast surgical oncologists and breast surgeons navigating adjuvant therapy decisions, the PREDICT Breast model has become an indispensable prognostication tool endorsed by major international clinical guidelines. At OncoToolkit, we've built a streamlined PREDICT Breast (Prognostic Index) calculator that computes the baseline prognostic index in seconds, eliminating manual calculations during busy multidisciplinary team (MDT) meetings and point-of-care consultations.
This article provides a comprehensive overview of the PREDICT Breast prognostic model, its clinical evidence base, and how our calculator can support both routine clinical care and research applications.
2. What Is the PREDICT Breast Prognostic Model?
PREDICT Breast is an online prognostication tool originally developed at the University of Cambridge in 2007 to help clinicians and patients make informed decisions about adjuvant treatment following surgery for early invasive breast cancer. Unlike simple staging systems such as the TNM classification, PREDICT integrates multiple clinicopathological variables to generate individualized survival probability estimates at 5, 10, and 15 years.
The model calculates a Prognostic Index (PI), a composite score derived from weighted contributions of key tumor and patient characteristics. A lower PI correlates with better survival probability, while a higher PI indicates worse prognosis. The tool stratifies patients into risk categories that directly inform discussions about the potential benefits of adjuvant chemotherapy, endocrine therapy, and trastuzumab.
The American Joint Committee on Cancer (AJCC) has officially endorsed PREDICT Breast, and it is referenced in major international guidelines including those from NICE (UK), reflecting its robust evidence base and clinical utility.
2.1 Input Variables
The PREDICT model incorporates eight core variables that collectively capture the biological behavior and anatomical extent of the disease:
- • Age at diagnosis (years): Older patients generally have better breast cancer-specific survival but higher competing mortality risks.
- • Tumor size (mm): Measured as the maximum invasive tumor diameter on histopathology.
- • Number of positive lymph nodes: The strongest traditional prognostic factor in breast cancer.
- • Tumor grade (Grade 1, 2, or 3): Reflects tumor differentiation and correlates with proliferative activity.
- • Estrogen receptor (ER) status (positive or negative): Determines eligibility for endocrine therapy and influences prognosis.
- • HER2 status (positive or negative): Identifies candidates for anti-HER2 targeted therapy and affects baseline prognosis.
- • Ki-67 status (high or low proliferation): A proliferation marker that refines prognostic estimates, particularly in ER-positive disease.
- • Mode of detection (screening-detected vs. symptomatic): Screen-detected cancers carry a survival advantage beyond stage shift alone.
This combination of anatomical, biological, and clinical factors enables PREDICT to provide more refined prognostic estimates than traditional staging systems alone. Notably, the inclusion of mode of detection is unique to PREDICT and addresses the observation that screen-detected breast cancers have improved survival even when matched for tumor size, grade, and nodal status—likely reflecting differences in underlying tumor biology.
3. Why PREDICT Breast Matters in Clinical Practice
Adjuvant chemotherapy decisions in early breast cancer are among the most consequential discussions breast surgeons have with patients. The challenge lies in quantifying the absolute benefit of additional treatment against its toxicities—a calculation that varies substantially based on individual patient and tumor characteristics. Without digital tools, this process is prone to cognitive overload, inconsistency, and potential treatment misallocation.
3.1 The Problem with Traditional Decision-Making
Before digital prognostication tools, clinicians relied on:
- Complex nomograms: Paper-based charts that were cumbersome to use in clinic and prone to reading errors.
- Mental estimation: Often inaccurate and highly variable between clinicians.
- Outdated models: Tools that did not incorporate contemporary variables like HER2 status or Ki-67.
- Population-based statistics: Applying median outcomes to individual patients, ignoring personal risk profiles.
3.2 Addressing "Calculator Fatigue" in Busy MDTs
PREDICT addresses this by providing transparent, reproducible survival estimates that can be generated in seconds. On our platform, this calculator is mobile-responsive and optimized for rapid use—whether you're preparing cases the night before an MDT or consulting at the bedside during ward rounds.
The Cambridge Breast Unit framework for absolute 10-year survival benefit:
- • Less than 3% benefit: Chemotherapy not recommended
- • 3-5% benefit: Discuss treatment options with patient
- • More than 5% benefit: Chemotherapy recommended
3.3 Guideline Endorsement and Global Adoption
In 2018, the UK National Institute for Health and Care Excellence (NICE) recommended PREDICT Breast as the preferred prognostication tool for planning adjuvant therapy in early and locally advanced breast cancer. NICE guidance specifies that PREDICT should be used prior to considering additional molecular profiling tests like Oncotype DX, making it the first-line decision support tool in the UK clinical pathway.
4. Clinical Evidence and Validation
4.1 The Mathematical Foundation
PREDICT Breast uses a Cox proportional hazards regression model to estimate breast cancer-specific mortality and other-cause mortality separately. This dual-endpoint approach is methodologically important because it accounts for the fact that older patients, while having better breast cancer outcomes, face higher risks of death from other causes.
PI = beta1(age) + beta2(size) + beta3(nodes) + beta4(grade) + beta5(ER) + beta6(HER2) + beta7(Ki67) + beta8(detection)
The baseline survivor function and coefficient estimates are applied to generate personalized survival curves. For clinicians interested in the technical details, the University of Cambridge publishes the full mathematical specification, though our calculator handles all computations automatically.
4.2 Version History and Updates
| Version | Key Improvements |
|---|---|
| 1.0 (2007) | Original model based on 5,694 patients. |
| 2.0 (2012) | Added HER2 status and trastuzumab benefit. |
| 2.1 (2017) | Incorporated Ki-67 as optional variable. |
| 3.0 (2024) | Radiotherapy benefits and treatment harms added. |
| 4.0 (2025) | Improved calibration for contemporary regimens. |
4.3 Validation Studies
UK validation cohorts demonstrated AUC statistics ranging from 0.74 to 0.81, indicating good discriminatory ability. The AUC for 15-year breast cancer survival reached 0.809 in the West Midlands validation dataset and 0.846 in pooled data from other English registries.
Chinese validation cohorts: A study of 5,424 patients from Wenzhou Medical University (2024) compared PREDICT versions 2.2 and 3.0. Version 3.0 showed significantly improved calibration, with predicted 5-year overall survival deviating from observed by only -2.0% for the entire cohort.
Dutch validation (2025): A recent comparative validation of PREDICT versions 3.1 and 2.2 in the Dutch breast cancer population confirmed that version 3.1 provides improved calibration for contemporary patients.
4.4 Known Limitations
Accuracy reduced in very young patients (< 35y) and elderly patients (> 75y) due to aggressive biology and competing risks.
Calibration is less reliable for very large tumors (> 50mm) or extensive nodal involvement (10+ positive nodes).
Not designed for patients who received chemotherapy before surgery; pathological findings do not directly correspond.
Outcomes may exceed predictions due to rapid advances in anti-HER2 targeted therapies.
5. How Our Calculator Works
5.1 Step 1: Enter Patient and Tumor Data
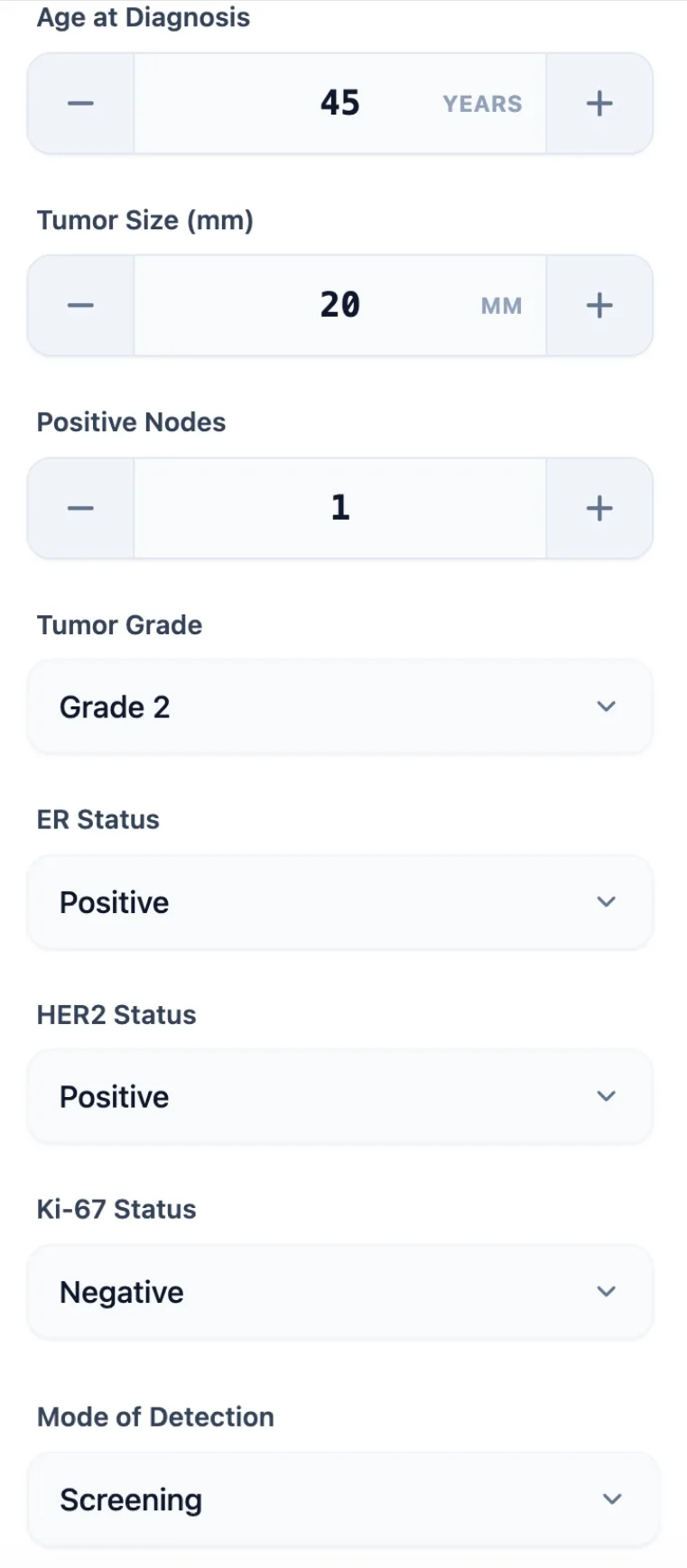
Figure 1. The input interface captures age, tumor size, nodal status, grade, receptor status, Ki-67, and detection mode—all variables needed to compute the Prognostic Index. The intuitive design minimizes data entry errors in time-pressured clinical settings.
5.2 Step 2: Review the Calculated Result
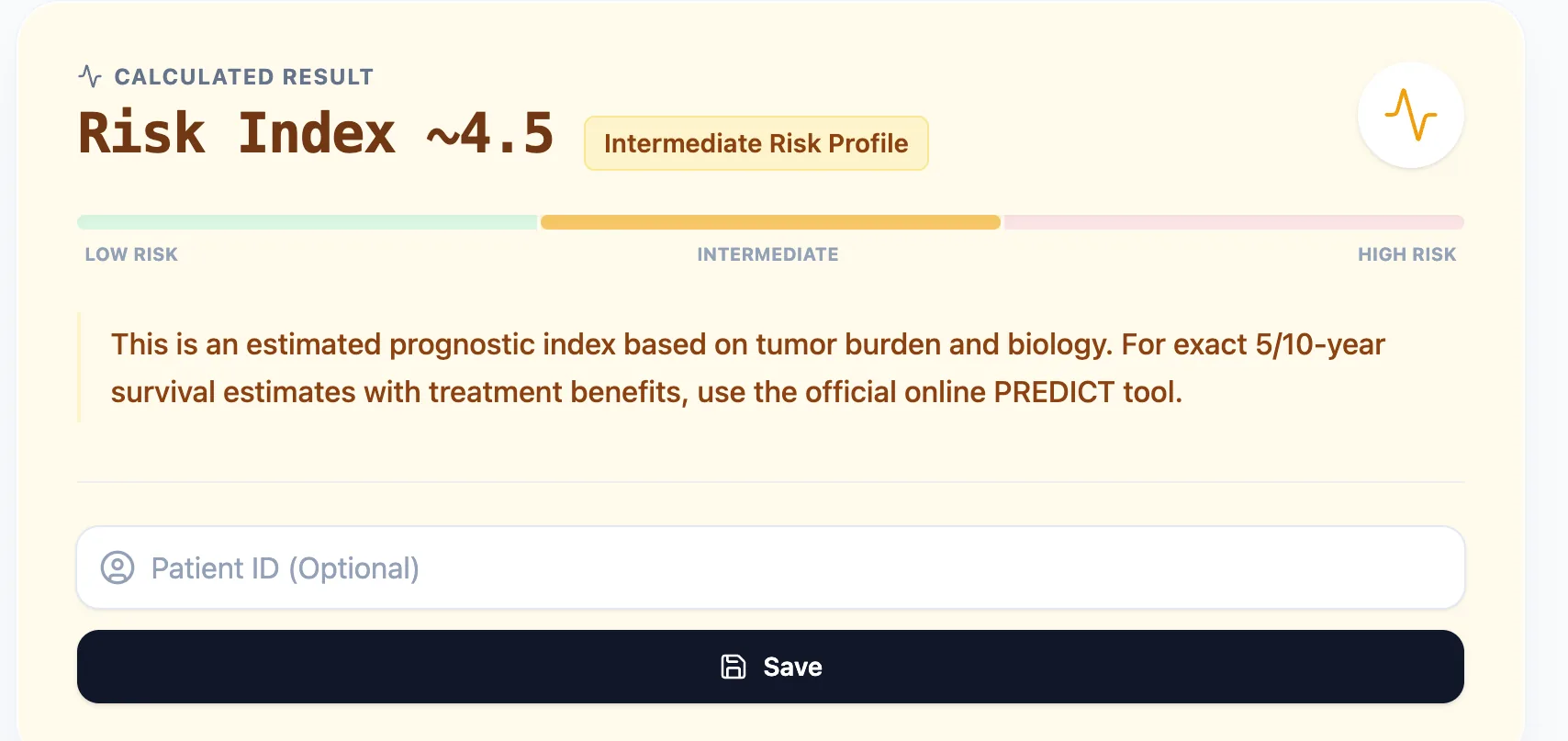
Figure 2. The result interface displays the Prognostic Index value and risk category with a visual scale, enabling quick communication of prognosis to patients and colleagues. The intermediate risk classification suggests this case warrants detailed discussion of adjuvant therapy options.
5.3 Step 3: Understand the Clinical Context
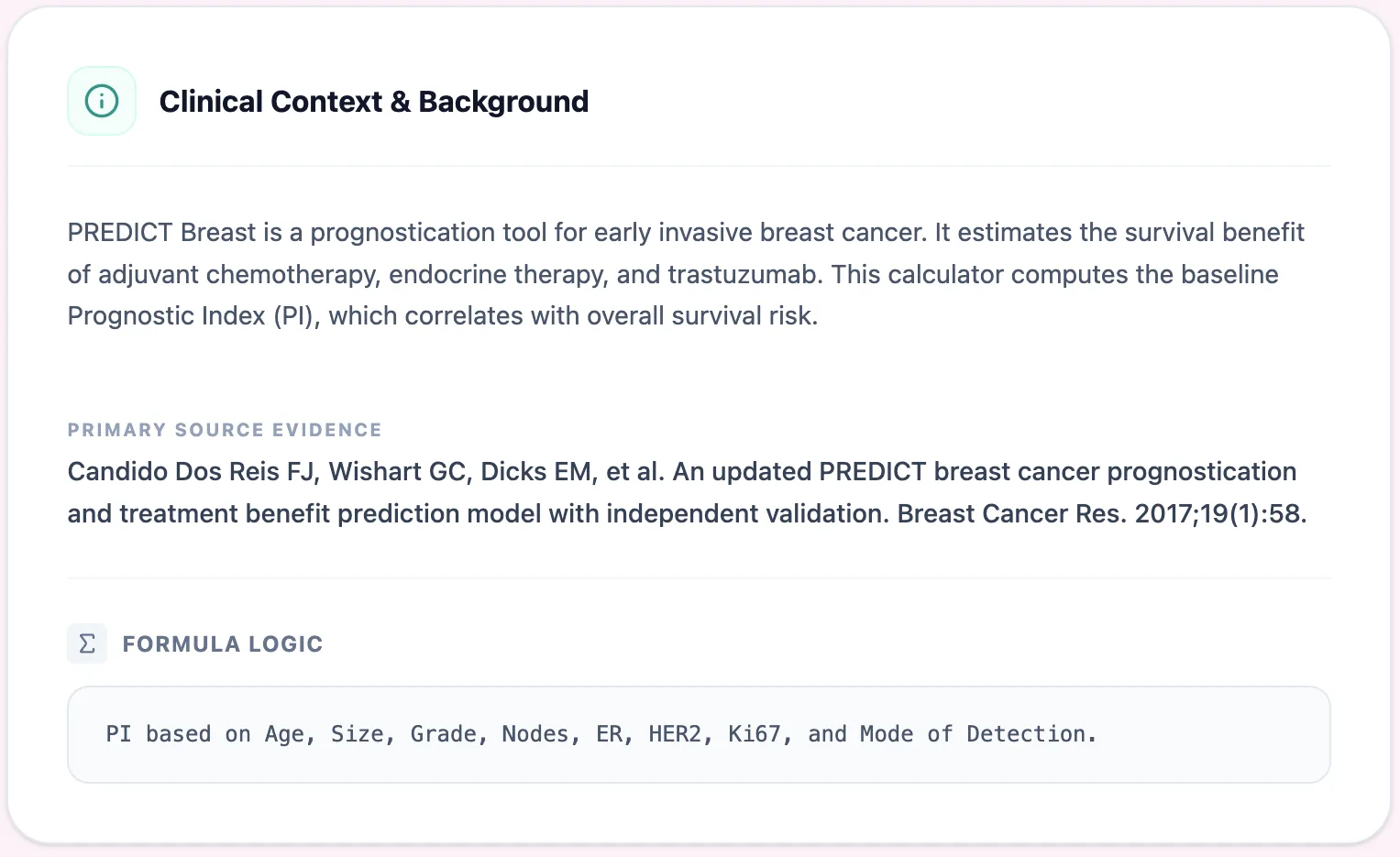
Figure 4. The clinical background section provides transparency about the tool's evidence base, primary source publication, and underlying formula logic, supporting informed use by clinicians and trainees.
5.4 Step 4: Save and Export
Figure 3. The reference data panel summarizes PI interpretation, reinforcing the inverse relationship between prognostic index and survival probability. This quick-reference guide supports accurate result interpretation.
6. Understanding the Variables in Detail
6.1 Mode of Detection: Why It Matters
One variable unique to PREDICT is mode of detection—whether the cancer was screen-detected or symptomatic. Research from the Eastern Cancer Registry demonstrated that screen-detected breast cancers carry a significant survival advantage beyond what can be explained by stage shift alone.
6.2 Ki-67: Interpreting Proliferation Status
Ki-67 is a nuclear protein expressed during active phases of the cell cycle, making it a useful proliferation marker. However, significant challenges exist with analytical validity and reproducibility, leading the International Ki-67 Working Group (IKWG) to suggest a 30% cutoff to denote high-risk tumors.
7. Supporting Clinical Care and Research
7.1 Routine Clinical Decision Support
The primary use case for our PREDICT Breast calculator is supporting adjuvant therapy discussions, including pre-MDT preparation, bedside consultations, and clinical documentation for audits.
7.2 Education and Training
For fellows and trainees, the calculator serves as an interactive tool for understanding how individual variables contribute to prognosis through case-based learning and sensitivity analysis.
7.3 Clinical Research and Quality Improvement
Our platform enables standardized baseline risk stratification for observational studies and longitudinal outcome tracking through saved patient records.
8. Comparison with Other Prognostic Tools
| Feature | NPI | PREDICT |
|---|---|---|
| Variables | 3 (size, nodes, grade) | 8 (includes biology & age) |
| Output | Risk group | Personalized probability |
| Treatment benefit | No | Yes (Multimodal) |
| Endorsement | Historical standard | NICE / AJCC endorsed |
9. Frequently Asked Questions
Can PREDICT be used in patients who received neoadjuvant chemotherapy?
No. The PREDICT model was developed and validated on patients who underwent primary surgery without prior systemic therapy. In neoadjuvant-treated patients, post-treatment pathological findings do not directly correspond to the variables the model interprets.How does PREDICT differ from Oncotype DX?
PREDICT is a prognostic model based on clinicopathological features, whereas Oncotype DX is a genomic assay specifically for ER-positive, HER2-negative patients to predict chemotherapy benefit.
What is the most common mistake when calculating the PREDICT score?
Frequent errors include entering tumor size in centimeters instead of millimeters and confusing total nodes examined with the number of positive nodes.
Ready to Streamline Your Prognostic Assessments?
Focus on meaningful patient conversations with our data-driven calculator.
Use the PREDICT Calculator
Free to use. No registration required.