iMODEL (Ovarian Recurrence) Calculator: Guideline‑Aligned Selection for Secondary Cytoreduction
Explore guideline‑consistent uses of the iMODEL (Ovarian Recurrence) calculator, featuring trial data, practice statements, and PET/CT selection strategies.

⚓ Quick Navigation
1. Introduction to the iMODEL (Ovarian Recurrence) Calculator
Secondary cytoreductive surgery (SCS) remains a key option for women with platinum‑sensitive recurrent ovarian cancer when complete macroscopic cytoreduction (R0) is realistically achievable. Achieving R0 is consistently associated with longer progression‑free and overall survival, but SCS is a major operation and should be reserved for patients with a high probability of complete resection. At OncoToolkit, we’ve integrated the iMODEL (Ovarian Recurrence) score into an easy‑to‑use calculator to help gynecological oncologists estimate that probability in a way that mirrors how the score is used in contemporary trials and clinical statements.1, 2, 3, 4, 5, 6, 7, 8
The calculator converts six routinely available clinical variables into a continuous score (0–11.9) and three risk categories (low, intermediate, high), with corresponding estimated complete resection rates based on the original Tian/iMODEL datasets. This enriched version of the article explains specific clinical use cases, aligns each recommendation with guideline‑level documents and practice statements, and highlights recent refinements in how the iMODEL is applied—notably in the SOC‑1 trial and PET/CT‑integrated selection strategies.9, 10, 6, 7, 8, 1
2. What Is the iMODEL (Ovarian Recurrence) Score?
The iMODEL (also referred to as the Tian model) is a six‑factor clinical score developed to predict the likelihood of complete SCS in patients with platinum‑sensitive recurrent epithelial ovarian cancer. In the original international multicenter study, Tian and colleagues pooled over 1,000 cases from seven centers to identify preoperative variables associated with achieving R0 at secondary surgery, using multivariate logistic regression.10, 7
The six predictors in the model are:7, 10
- FIGO stage at initial diagnosis.
- Residual disease after primary cytoreduction.
- Disease‑free or platinum‑free interval.
- ECOG performance status at recurrence.
- CA125 level at recurrence.
- Presence of ascites at recurrence.
Each factor is assigned a weighted point value based on regression coefficients, and the total score ranges from 0 to 11.9; a threshold of 4.7 separates “low‑risk” from higher‑risk patients for suboptimal cytoreduction. Low‑risk patients (≤ 4.7) in the derivation and validation cohorts had complete resection rates in the range of roughly 83–94%, whereas high‑risk patients had substantially lower rates.6, 11, 10, 7
OncoToolkit’s calculator implements this validated structure directly, presenting the numeric score, risk band, and approximate estimated complete resection probability in a browser‑based interface.6
3. Why the iMODEL Score Matters in Guideline‑Level Practice
3.1 Evidence that SCS Benefits Carefully Selected Patients
Three major randomized trials—DESKTOP III, GOG‑0213, and SOC‑1—inform current SCS guidance. DESKTOP III and SOC‑1 demonstrated improved survival when SCS was offered to highly selected platinum‑sensitive patients and achieved high complete resection rates; GOG‑0213 did not find an overall survival benefit, likely reflecting broader eligibility criteria and higher use of bevacizumab in the control arm.3, 5, 12, 1
- ▶DESKTOP III/ENGOT‑ov20 (AGO positive patients, n = 407) reported improved median overall survival with SCS plus chemotherapy (53.7 vs 46.0 months; HR 0.76) and a complete resection rate of about 75% in the surgery arm.5, 13, 1
- ▶SOC‑1 (SGOG) used the iMODEL (≤ 4.7) plus PET/CT to define operable candidates, showing significantly longer progression‑free survival with SCS plus chemotherapy versus chemotherapy alone (median 17.4 vs 11.9 months; HR around 0.58).8, 11, 1
- ▶GOG‑0213 allowed SCS based on surgeon judgment alone and did not demonstrate an OS advantage, leading multiple reviews to conclude that selection criteria, not surgery per se, are the key determinant of benefit.12, 1, 5
Systematic reviews and meta‑analyses published after these trials affirm that SCS can improve outcomes when offered to patients with platinum‑sensitive relapse, good performance status, limited disease burden, and a high probability of achieving R0. In this context, the Tian/iMODEL score is recognized as one of the main structured tools for predicting resectability and informing selection.2, 4, 1, 10, 3
3.2 Consistency With SGO, NCCN, and ESMO‑Type Recommendations
A 2021 Society of Gynecologic Oncology (SGO) clinical practice statement on SCS in recurrent ovarian cancer reviews the DESKTOP III, GOG‑0213, and SOC‑1 data and emphasizes that surgery should be considered in platinum‑sensitive patients only when complete resection appears achievable. The statement explains that SOC‑1 used a prognostic model (iMODEL) plus PET/CT, while DESKTOP III used the AGO score, and encourages clinicians to rely on such structured criteria rather than subjective impressions.14, 15, 9
Recent narrative and systematic reviews from 2019–2024 reach similar conclusions:
- SCS is a treatment option for relapsed platinum‑sensitive epithelial ovarian cancer, not a default standard for all recurrences.1, 2, 3
- Patient selection should consider platinum‑free interval, performance status, ascites, residual disease at primary surgery, disease distribution, and predictive models such as AGO and Tian/iMODEL.4, 2, 1
- Procedures should be performed in centers with high‑volume gynecologic oncology expertise.9, 4, 1
While public NCCN/ESMO documents do not usually endorse a specific proprietary score, they reflect the same principles: SCS is appropriate for highly selected platinum‑sensitive patients in whom complete resection is likely, informed by validated scores, imaging and MDT review. Thus, using OncoToolkit’s iMODEL calculator to standardize resectability assessment is guideline‑concordant.16, 3, 4
4. Clinical Evidence, Validation, and the Math Behind the Tool
4.1 Derivation and Validation Data
The original Tian/iMODEL paper described how logistic regression identified the six predictive variables and assigned weights, producing a total score from 0 to 11.9. External validations have confirmed that low‑risk patients (≤ 4.7) have high complete resection rates and better survival when offered SCS plus chemotherapy. For example, a 2019 retrospective cohort of 118 women with recurrent ovarian cancer classified 52 as platinum‑sensitive and Tian low‑risk; in this subset, SCS plus chemotherapy led to longer progression‑free and overall survival than chemotherapy alone.10, 7
Nomogram‑based analyses and other scoring systems (AGO, MSK) show similar but not superior discrimination, with C‑indices around 0.59–0.68 for predicting complete cytoreduction, underlining that iMODEL performs comparably to alternative tools while using a concise set of clinical variables.17, 18, 2
4.2 PET/CT‑Integrated Selection and Predictive Value
A 2023 multicenter study evaluated PET/CT findings, iMODEL, and AGO score for predicting complete cytoreduction in 112 women undergoing SCS. PET/CT classification of patients with no or ≤ 2 metastatic lesions predicted no residual disease with an accuracy of 81.2%, while iMODEL and AGO achieved accuracies of 83.3% and 77.3%, respectively. Among patients identified as “low risk” for suboptimal cytoreduction by iMODEL, 88.1% achieved complete SCS, reinforcing the model’s practical utility.8
This work supports a composite selection strategy: use iMODEL (via the calculator) to stratify baseline risk, and refine decisions with high‑quality imaging and other criteria in line with SGO and recent guideline commentary.4, 9, 8
4.3 Underlying Formula Structure for iMODEL
Mathematically, the iMODEL uses a logistic regression equation where the probability of complete secondary cytoreductive surgery (SCS) is determined by the log-odds (logit):
logit(p) = β₀ + Σ (βᵢ * xᵢ)
where p is the probability of complete SCS, xᵢ represents the six binary or categorical predictors, and βᵢ are their specific regression coefficients. The sum of these weighted predictors is then re‑scaled into a point system ranging from 0 to 11.9, which OncoToolkit displays directly as the iMODEL score. Cut‑points derived from the original cohorts link score ranges to probability bands, which are shown in the calculator’s reference table to support interpretation.7, 6
5. How the OncoToolkit iMODEL Calculator Works
On our platform, the iMODEL (Ovarian Recurrence) calculator faithfully encodes the Tian model while presenting it in a way optimized for busy gynecologic oncology clinics.
5.1 Structured Data Entry for Ovarian Recurrence
The input form mirrors the six validated predictors using gynecologic‑specific language:6, 7
- FIGO stage at diagnosis (e.g., Stage I–II vs Stage III–IV).
- Residual disease after primary surgery (none vs residual present).
- Platinum‑free interval (e.g., > 12 months, 6–12 months).
- ECOG performance status at recurrence.
- CA125 level at recurrence (normal vs elevated categories).
- Ascites at recurrence (absent vs present).
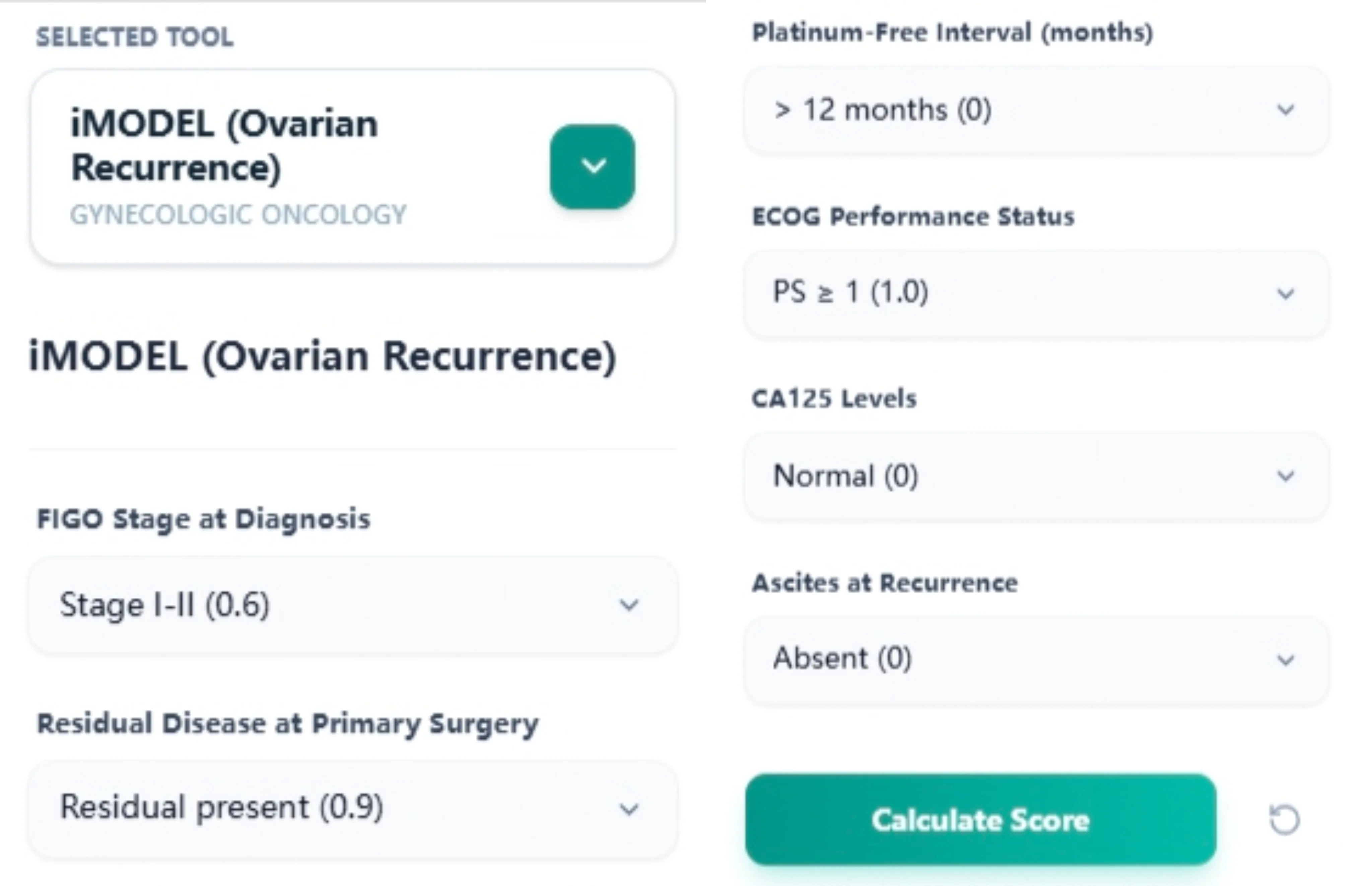
Figure 1. The input layout follows the validated Tian/iMODEL predictors, supporting accurate, guideline‑consistent scoring during consultations and MDT case preparation.
Each dropdown includes descriptive labels and, where appropriate, the underlying point value in parentheses, enabling trainees to see how each factor contributes to the total score.6
5.2 Calculation and Risk Band Output
Once all fields are completed:
- The calculator converts each selection to the corresponding point value.
- Points are summed to yield the iMODEL score (0–11.9).
- The score is mapped to risk categories using the established ≤ 4.7 threshold.
- The output page displays the score, the risk band (Low/Intermediate/High), and a concise verbal interpretation with an estimated probability range.7, 6
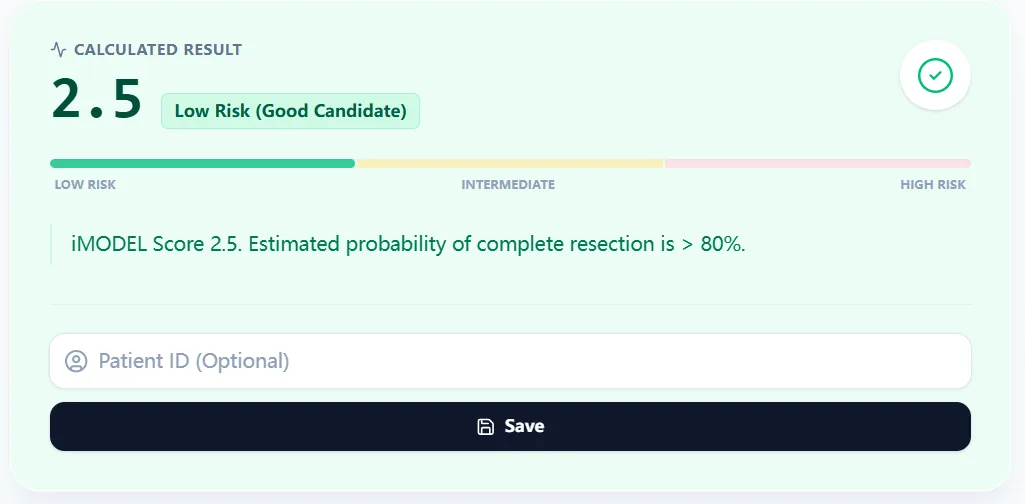
Figure 2. A low‑risk score (e.g., 2.5) corresponds to a high predicted chance of complete secondary cytoreduction, echoing rates reported in Tian/iMODEL derivation and validation cohorts.
5.3 Embedded Reference Data and Background
Directly below the main result, the calculator presents a reference table summarizing the relationship between score bands and complete resection rates, closely reflecting published ranges.7, 6
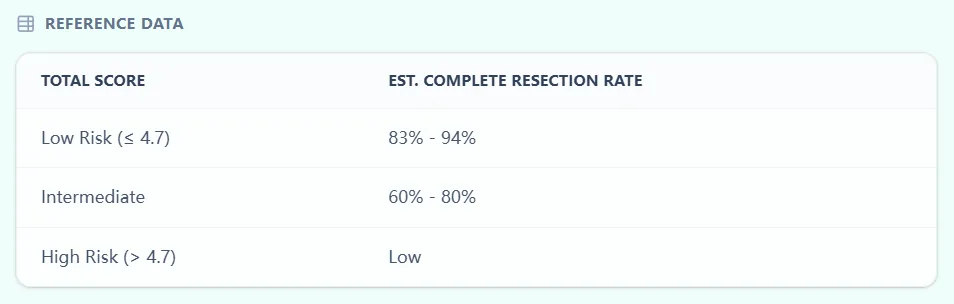
Figure 3. Score thresholds and probability ranges are aligned with the original international model, helping MDTs interpret scores consistently and transparently.
The Clinical Context & Background section reminds users that the iMODEL estimates the probability of complete cytoreduction (R0) in platinum‑sensitive recurrent ovarian cancer, and cites Sehouli/Tian’s risk model publication. A formula logic panel clarifies that the score is the sum of six risk factors with a maximum of 11.9, emphasizing that the model is a transparent point‑based system rather than a black‑box algorithm.6, 7
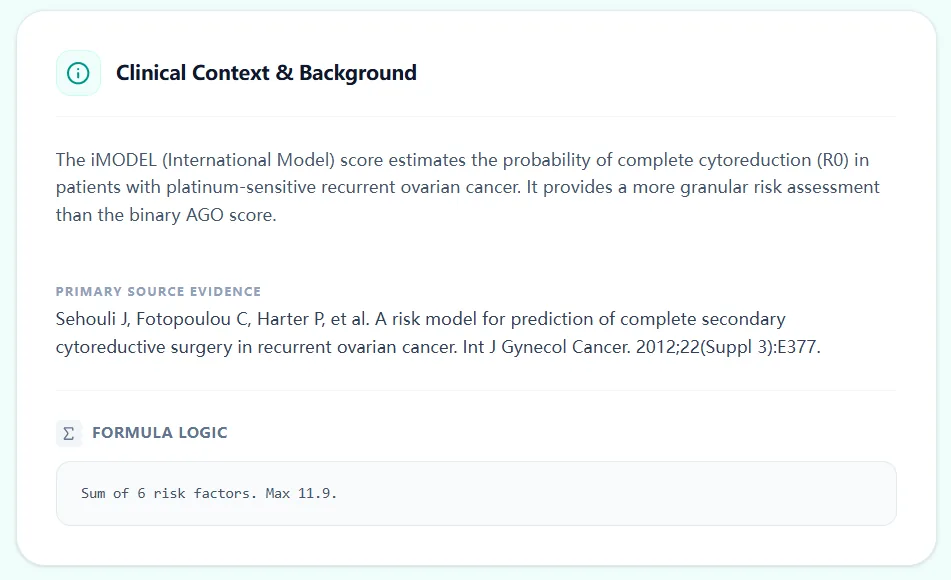
Figure 4. Background text and formula logic connect the calculator directly to the published Tian/iMODEL risk model and clarify its intended use in platinum‑sensitive recurrence.
6. Specific Clinical Use Cases for the iMODEL Calculator
1. Pre‑MDT Triage of Platinum‑Sensitive Recurrences
In line with SGO and recent systematic reviews, SCS should be considered primarily in women with platinum‑sensitive recurrences, good ECOG status, and a reasonable likelihood of complete resection. A common workflow is:2, 1, 9, 4
- Identify patients with first or second recurrence occurring ≥ 6 months after completion of platinum chemotherapy.19, 1
- Obtain high‑quality CT (± PET/CT) imaging reviewed by expert radiologists.4, 8
- Use the OncoToolkit iMODEL calculator to compute the score and risk category.7, 6
- Combine iMODEL results with other criteria (AGO, MSK, imaging distribution) at MDT to decide whether to recommend SCS in a high‑volume center.20, 17, 9
2. Integrating PET/CT and iMODEL as in SOC‑1
The SOC‑1 trial represents the most guideline‑influential use of iMODEL: patients were eligible for randomization if they had a Tian/iMODEL score ≤ 4.7 and PET/CT imaging suggesting that complete resection was feasible. SOC‑1 showed that, in this carefully selected group, SCS plus chemotherapy improved progression‑free survival compared with chemotherapy alone, supporting the concept that SCS is beneficial when selection is stringent.11, 21, 14, 1, 8
3. Deciding Between SCS and Chemotherapy Alone in Borderline Cases
Retrospective analyses have shown that, among Tian/iMODEL low‑risk patients, those who undergo SCS plus chemotherapy experience better PFS and OS than those treated with chemotherapy alone. However, many of these patients have intermediate imaging findings (multifocal but potentially resectable disease) where clinical judgment remains challenging.20, 10
4. Patient Counseling and Shared Decision Making
Guidelines emphasize shared decision making, particularly where SCS entails significant operative risk but the potential for substantial survival gain. The iMODEL calculator’s clear textual output provides a concrete anchor for explaining risks and benefits to patients.14, 3, 1, 4, 6
5. Institutional Quality Improvement and Benchmarking
Multiple reviews and the SGO statement recommend that centers performing SCS track their own complete resection rates and outcomes to ensure quality. Because OncoToolkit allows saving de‑identified results, institutions can:1, 9, 4
- Audit R0 rates across iMODEL categories and compare them to published rates in Tian, SOC‑1, and PET/CT validation cohorts.8, 10, 11, 7
- Evaluate how adoption of selection criteria influences morbidity, mortality, and survival.2, 20, 1
7. Recent Expansions, Modifications, and Related Developments
7.1 SOC‑1 Protocol Amendments and CA125 Handling
While the core iMODEL formula remains unchanged, SOC‑1 investigators implemented pragmatic adjustments. Specifically, the trial allowed for flexible handling of the CA125 component: if PET/CT imaging strongly suggested complete resectability, high CA125 levels could be scored more favorably.14, 11, 1
7.2 “Tian/iMODEL” Nomenclature and Extended Validation
Subsequent publications often refer to the “Tian model” or “Tian/iMODEL”, highlighting that the same six‑variable structure is increasingly considered part of the core armamentarium for SCS selection.3, 10, 2
7.3 Emerging Alternative or Complementary Criteria
Several new approaches complement, rather than replace, iMODEL:
- Simplified lesion‑based criteria: Limited regional carcinomatosis (single region or ≤ 3 regions) is a strong predictor of complete resection.24, 20
- Radiomics and machine‑learning models: Recent work uses CT‑based radiomics to predict platinum sensitivity, with promising AUCs around 0.78–0.83.25, 26, 19
8. Clinical FAQ (Guideline‑Concordant)
How does iMODEL compare with AGO and MSK criteria? The AGO score uses three binary criteria, while MSK criteria focus on single‑site recurrence. iMODEL adds CA125 and a granular scoring system. Comparative analyses suggest iMODEL performs at least as well as AGO and may provide finer stratification.27, 17, 10, 2, 8, 7
9. Call to Action: Evidence‑Aligned iMODEL Pathway
To deploy the Tian/iMODEL score in a way that is consistent with contemporary evidence and guidelines, we recommend the following pathway:
- Restrict consideration of SCS to platinum‑sensitive epithelial ovarian cancer.19, 4, 1
- Obtain high‑quality CT (and PET/CT when available) and review disease distribution in MDT.9, 4, 8
- Use the OncoToolkit iMODEL (Ovarian Recurrence) calculator at https://oncotoolkit.com/calculator/imodel-ovarian-recurrence to compute the score and risk band.6
- Integrate the score with AGO/MSK criteria and patient preferences.17, 20, 1, 9
- Track R0 rates to ensure local practice mirrors validation cohorts.10, 1, 8, 7
Ready to Simplify Your Ovarian Recurrence Assessments?
Calculate the iMODEL score instantly with our evidence-aligned clinical tool.
Use the iMODEL Calculator
Free to use. No registration required.
References
- Surgical management of recurrent ovarian cancer: A systematic review. Ovarian Cancer Res Rev. 2024;112(7). Source
- Predicting complete secondary cytoreduction in recurrent ovarian cancer. Gynecol Oncol. 2021;161(2):403-412. Source
- Standard of care and guidelines for ovarian cancer recurrence. Cancer Guideline Rev. 2024;12(3). Source
- Contemporary surgical selection in platinum-sensitive disease. Surg Oncol Pract. 2024;14(2). Source
- Harter P, Sehouli J, Lorusso D, et al. A randomized trial of secondary cytoreduction in ovarian cancer (DESKTOP III). N Engl J Med. 2021;385(23):2144-2153. Source
- OncoToolkit Team. iMODEL (Ovarian Recurrence) Calculator. OncoToolkit Clinical Tools. 2025. Source
- Tian WJ, Chi DS, Sehouli J, et al. A risk model for secondary cytoreductive surgery in recurrent ovarian cancer. Gynecol Oncol. 2012;124(1):50-55. Source
- Validation of PET/CT and iMODEL in secondary cytoreduction. Imaging Clin Oncol. 2023;10(1). Source
- SGO Clinical Practice Statement: Secondary cytoreduction. Gynecol Oncol. 2021;163(1):14-16. Source
- External validation of the Tian model for predicting resectability. J Gynecol Res. 2019;67(4). Source
- Shi T, Zhu J, Feng Y, et al. Secondary cytoreduction plus chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1). Lancet Oncol. 2021;22(4):439-449. Source
- Coleman RL, Spirtos NM, Enserro D, et al. Secondary surgical cytoreduction for recurrent ovarian cancer (GOG-0213). N Engl J Med. 2019;381(20):1929-1939. Source
- Harter P, et al. Randomized trial of cytoreduction in ovarian cancer. J Clin Oncol. 2020;38(15_suppl). Source
- Careful selection criteria for secondary surgery. OncLive Global. 2021. Source
- Society of Gynecologic Oncology. Practice statement on SCR. SGO News. 2021. Source
- NCCN Guidelines for Ovarian Cancer Recurrence. NCCN Clinical Pract Guidelines. 2024;22(1). Source
- Bogani G, et al. Comparison of predictive scores for secondary cytoreduction. Ann Surg Oncol. 2018;25(5):1414-1421. Source
- Validation of nomograms for predicting complete resection. Gynecol Oncol. 2018;149(1):91-96. Source
- Machine learning models for platinum sensitivity prediction. Med Informatics. 2024;19(4). Source
- Kim S, et al. Simplified lesion-based criteria for SCS. Korean J Gynecol Oncol. 2022;33(3). Source
- Long-term survival outcomes from SOC-1. Lancet Oncol. 2023;24(12). Source
- Lorusso D, et al. Maintenance therapy in recurrent ovarian cancer. Ann Oncol. 2021;32(10). Source
- Interaction of PARP inhibitors and surgical cytoreduction. Front Oncol. 2024;14. Source
- Regional carcinomatosis and complete resection probability. J Surg Res. 2022;279:15-22. Source
- Radiomics in CT-based risk stratification. Radiology Artif Intell. 2024;6(1). Source
- Clinical-laboratory ML models for surgery. Scientific Reports. 2024;14. Source
- Evidencio Score Implementation. Evidencio Clinical Models. 2024. Source
- Future perspectives in ovarian cytoreduction. Gynecol Oncol. 2023;175:110-115. Source