1. Introduction to Renal Cell Carcinoma (RCC) Prognosis
Prognostication in advanced renal cell carcinoma is increasingly complex, especially as systemic options expand and more patients present with locally advanced or metastatic disease. Risk models such as IMDC (Heng) and MSKCC (Motzer) remain central to how nephrologists and oncologists individualize therapy, yet calculating and comparing these scores manually can be time‑consuming during busy clinics or tumor boards. At OncoToolkit, we’ve built the Renal Cell Carcinoma (RCC) Prognosis Panel calculator to bring both models into a single, streamlined, guideline‑aligned interface that delivers interpretable results in seconds at the point of care (https://oncotoolkit.com/calculator/panel-renal-risk).1, 2, 3
2. What is the RCC Prognosis Panel?
The RCC Prognosis Panel is a unified digital implementation of the IMDC (Heng) and MSKCC (Motzer) risk models for patients with advanced or metastatic RCC who are being considered for systemic therapy, including IO/TKI combinations. The panel quantifies the number of adverse prognostic factors and classifies patients into favorable, intermediate, or poor‑risk groups, each associated with characteristic median overall survival estimates derived from validation cohorts.2, 4, 1
The IMDC model uses six adverse factors: Karnofsky performance status (KPS) less than 80%, time from diagnosis to systemic therapy under one year, anemia, hypercalcemia, neutrophilia, and thrombocytosis. The MSKCC model incorporates five core variables—low KPS, short interval from diagnosis to treatment, anemia, hypercalcemia, and elevated LDH—with later extensions adding metastatic burden and prior radiotherapy. On our platform, these parallel frameworks are calculated simultaneously so clinicians can see how contemporary and historical risk systems align for the same patient.5, 6, 3, 4, 1
3. Why IMDC and MSKCC Risk Matter in Practice
For patients with locally advanced or metastatic RCC, risk group assignment is tightly linked to systemic treatment selection, prognosis counseling, and clinical trial eligibility. NCCN and European guidelines explicitly recommend IMDC risk stratification when choosing immune checkpoint inhibitor (ICI)–TKI or ICI–ICI combinations, particularly for clear‑cell RCC.7, 2
Risk category informs decisions such as using doublet IO (e.g., nivolumab–ipilimumab) in intermediate/poor‑risk disease, versus VEGFR‑TKI/ICI combinations across all risk groups, or VEGFR‑TKI monotherapy in selected favorable‑risk patients.
Without digital support, clinicians must mentally track six IMDC and five MSKCC factors, cross‑reference tables of survival outcomes, and reconcile subtle differences between models—tasks that are error‑prone and add cognitive load in MDT discussions. On our platform, this calculator is mobile‑responsive and optimized for use during nephrology clinics, virtual MDTs, or bedside consults, helping teams standardize risk assessment while reducing “calculator fatigue.”8, 1, 2, 7
4. Clinical Context, Logic, and Risk Cut‑offs

Figure 1. Overview screen summarizing IMDC and MSKCC criteria.
4.1 IMDC (Heng) model
The IMDC model was developed from a large, international database of metastatic RCC patients treated with VEGFR‑targeted agents and later validated in cohorts receiving IO/IO and IO/TKI regimens. Adverse factors are:9, 3, 1
- KPS < 80%
- Time from diagnosis to systemic therapy < 1 year
- Hemoglobin below lower limit of normal
- Corrected calcium above upper limit of normal
- Neutrophil count above upper limit of normal
- Platelet count above upper limit of normal3, 1
Patients with 0 factors are favorable risk, 1–2 factors intermediate, and ≥3 factors poor risk; median overall survival historically ranged from roughly 43 months in favorable to under 8 months in poor‑risk groups in targeted‑therapy era cohorts.1, 2
4.2 MSKCC (Motzer) model and extensions
The MSKCC model predates targeted therapy and was derived from cytokine‑era clinical trials, identifying low KPS, high LDH, low hemoglobin, hypercalcemia, and short time from diagnosis to treatment as key predictors of survival. Original data showed median survival of approximately 28–30 months for favorable, 14–15 months for intermediate, and 4–5 months for poor‑risk disease, with expanded versions incorporating metastatic burden yielding updated survival estimates around 26, 14, and 7 months. On our panel, MSKCC is presented alongside IMDC primarily for historical comparison, trial stratification context, and for cases where clinicians wish to align with legacy datasets.10, 6, 4, 5
4.3 Formula logic
Both IMDC and MSKCC are point‑based additive models rather than regression formulas. Each adverse factor contributes one point; risk category is determined purely by the count of present factors. OncoToolkit’s RCC Prognosis Panel reflects this logic transparently by displaying the number of factors for each system and mapping 0, 1–2, and ≥3 factors to favorable, intermediate, and poor‑risk strata, respectively, along with associated historical median OS values.4, 1
5. Evidence and Validation Across Regions
The IMDC criteria have been validated across multiple continents, including North American, European, and Asian cohorts. In a meta‑analytic evaluation, IMDC risk groups showed consistent hazard ratios for overall survival when comparing intermediate versus favorable and poor versus favorable risk, with subgroup analyses confirming applicability in both Asian and non‑Asian populations.11, 9, 8
In Japanese and broader East Asian series of metastatic RCC treated with targeted therapy, IMDC classification remained strongly associated with overall and progression‑free survival, supporting its use in Asian nephrology and oncology practice. MSKCC criteria have similarly been validated and extended in Western trial populations, with additional factors such as prior radiotherapy and sites of metastasis refining risk discrimination. While MSKCC has also been applied in Asian datasets, contemporary guidelines now favor IMDC as the primary risk model in both Western and Asian practice, particularly in the IO/TKI era.6, 11, 5, 8, 2, 7, 4
6. Guideline Alignment and Recent Advances
Current NCCN Kidney Cancer Guidelines and European Association of Urology/ESMO recommendations endorse IMDC risk stratification as a key step before first‑line systemic therapy selection in advanced RCC. These guidelines integrate IMDC categories into treatment algorithms for IO/IO and IO/TKI combinations, differentiating, for example, when nivolumab–ipilimumab is reserved for intermediate/poor‑risk patients versus when IO/TKI combinations can be used across all risk groups.2, 7
7. How the RCC Prognosis Panel Calculator Works
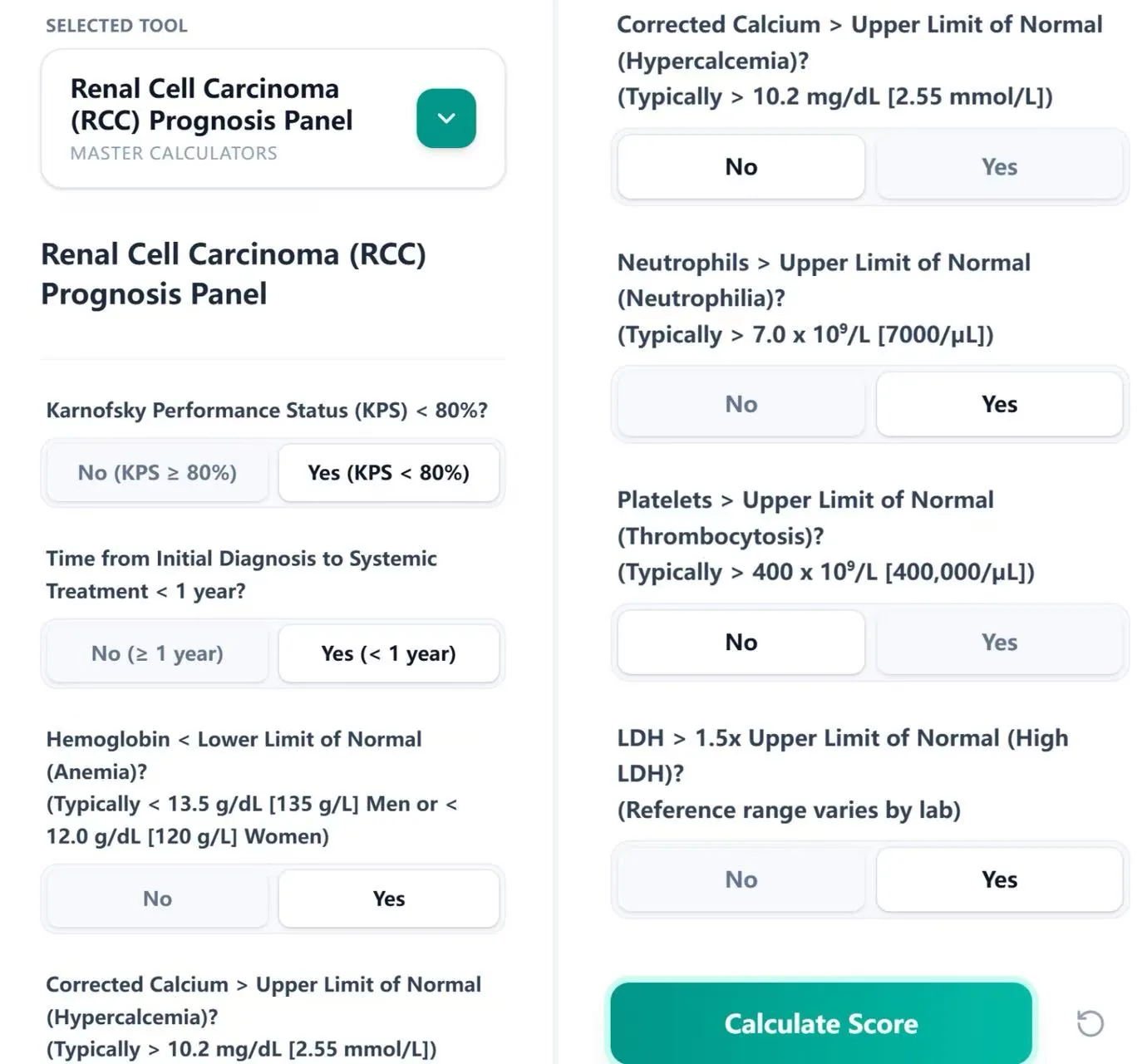
Figure 2. Input form for IMDC and MSKCC risk groups.
Using the calculator at https://oncotoolkit.com/calculator/panel-renal-risk, clinicians enter a small set of binary clinical and laboratory variables corresponding to the six IMDC and five MSKCC criteria. Inputs include Karnofsky performance status, interval from diagnosis to systemic therapy, hemoglobin level, corrected calcium, neutrophil count, platelet count, and LDH, with embedded hints referencing typical laboratory cut‑offs and the need to use local reference ranges.3, 1
8. Reference Data and Survival Context
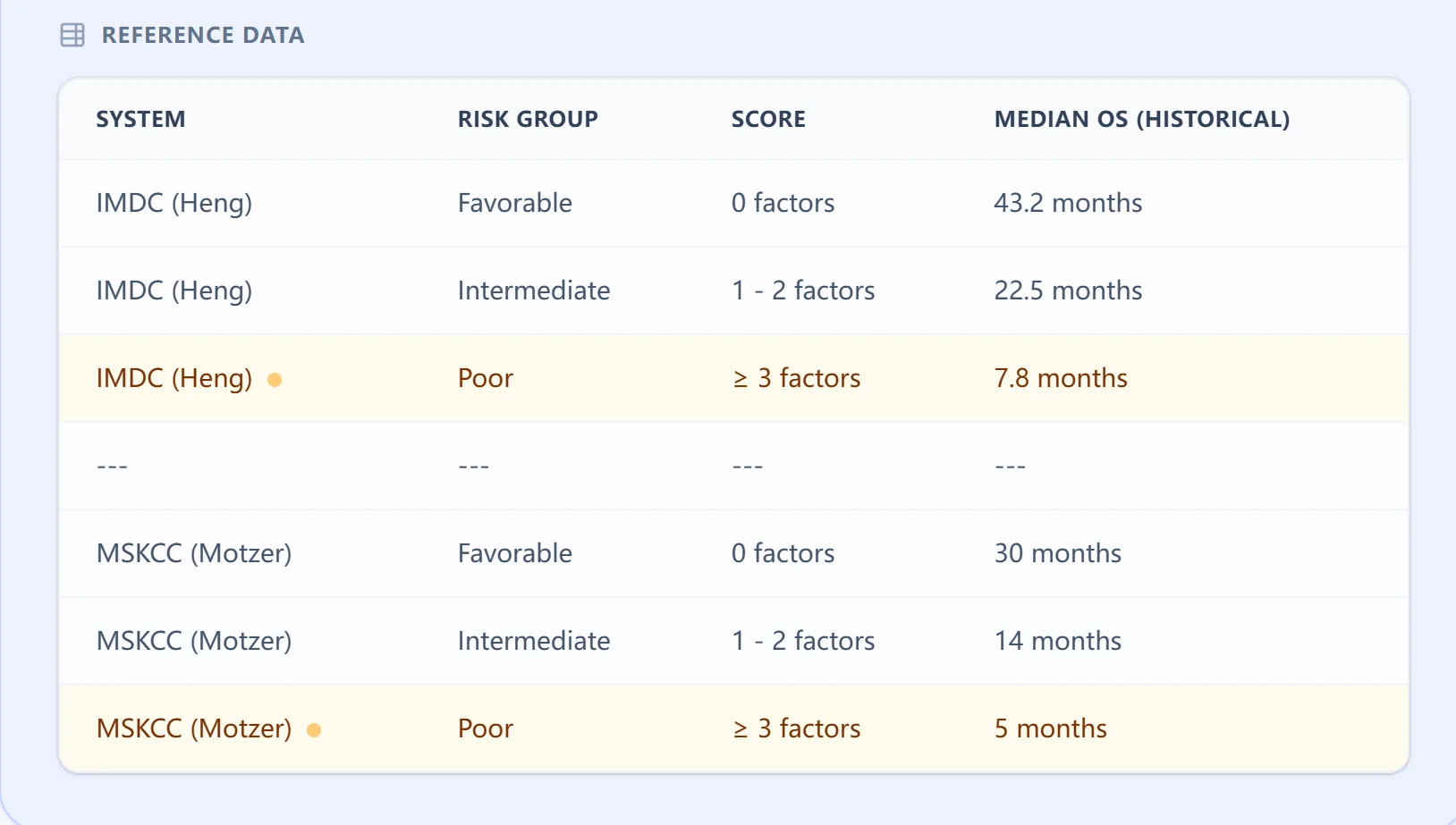
Figure 3. Reference table mapping risk factors to survival.
The reference table within the calculator summarizes risk group definitions and historical median overall survival for both IMDC and MSKCC systems. IMDC favorable‑risk patients (0 factors) are associated with multi‑year median OS, intermediate risk (1–2 factors) with shorter but still substantial survival, and poor‑risk (≥3 factors) with markedly reduced median OS in targeted‑therapy era cohorts. MSKCC categories show shorter historical survival times, reflecting earlier treatment eras and emphasizing why IMDC is preferred when counseling patients receiving modern IO/TKI combinations.10, 6, 5, 1, 2
9. Unified Panel Output and Interpretation
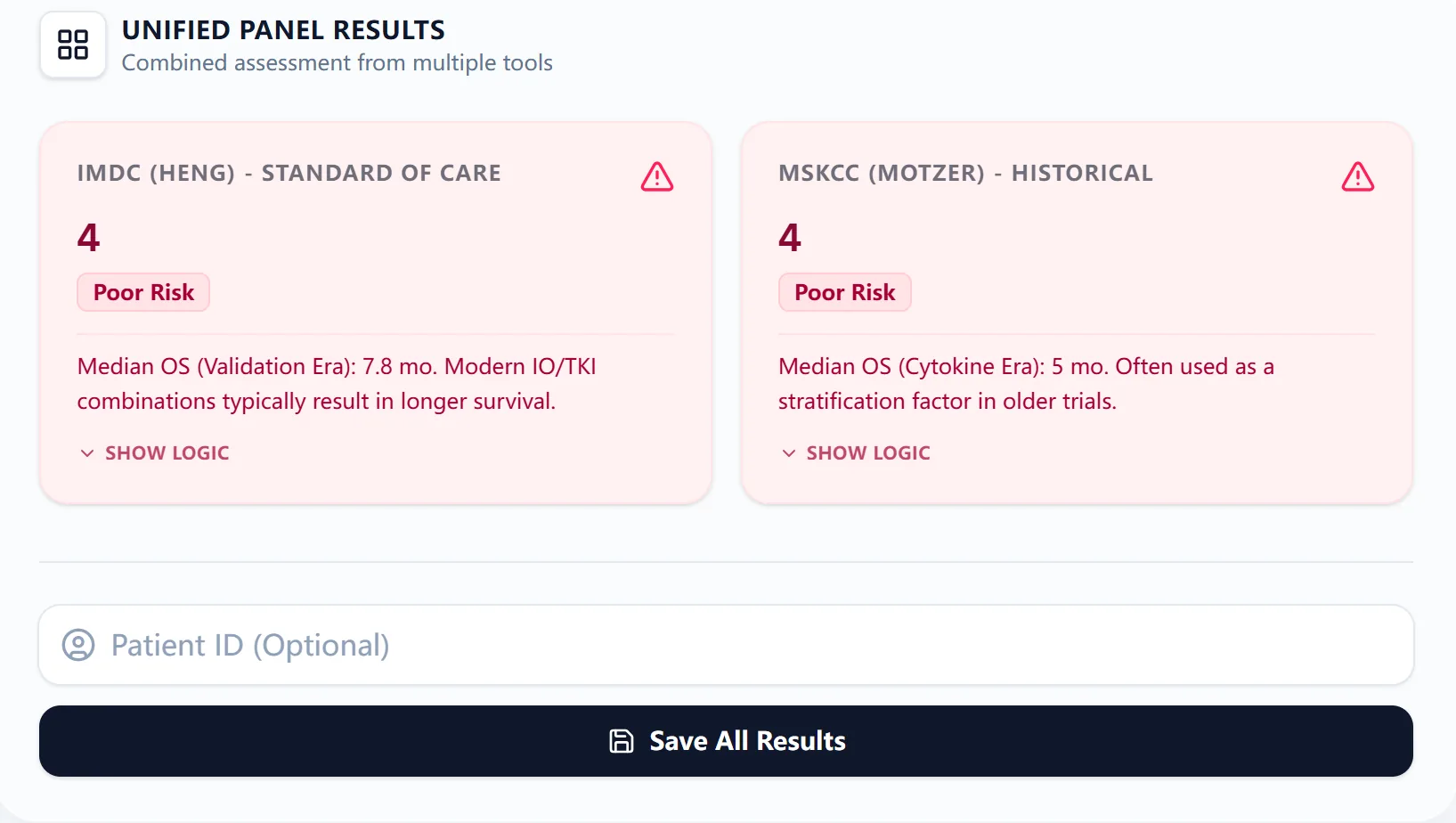
Figure 4. Example of unified panel results.
The unified results card presents IMDC and MSKCC outputs side‑by‑side, each with:
- The total number of adverse factors
- Risk label (favorable, intermediate, poor)
- Associated median OS based on historical validation data
- A brief interpretive statement about how modern IO/TKI combinations may alter outcomes in current practice1, 2
10. How OncoToolkit Supports Clinical Care and Research
At OncoToolkit, this calculator is designed to integrate into everyday nephrology and oncology workflows. Clinicians can use the RCC Prognosis Panel to:
- Rapidly assign IMDC risk at diagnosis of locally advanced or metastatic RCC to guide first‑line IO/TKI strategy.
- Standardize risk assessment ahead of MDT tumor boards, ensuring all team members are working from the same prognostic framework.
- Recalculate risk at key milestones (e.g., before switching systemic lines) when performance status or lab parameters change.
11. Specific Use Cases of the RCC Prognosis Panel
Clinically, nephrologists and oncologists can apply the tool in several common scenarios:
- Initial risk stratification for de novo metastatic RCC – Use the panel at first presentation to assign IMDC risk and select among IO/TKI combinations recommended by NCCN/EAU/ESMO for each category.7, 2
- Post‑nephrectomy recurrence or progression – In patients with prior localized disease who relapse, update risk classification using current labs and performance status before starting systemic therapy.
- Borderline cases between models – When IMDC and MSKCC differ (e.g., IMDC intermediate but MSKCC poor), use side‑by‑side results to frame nuanced discussions about prognosis and clinical trial fit.
- Cross‑regional practice – For centers caring for both Western and Asian patients, the tool’s alignment with validation data from multiple regions supports confident use across diverse populations.9, 11, 8
12. Regional Validation: Western vs Asian Populations
Several studies have evaluated IMDC and MSKCC performance across geographic regions. In international datasets, IMDC risk groups show similar hazard ratios for overall survival when comparing intermediate or poor‑risk to favorable‑risk patients, regardless of Asian versus non‑Asian origin. Japanese and other Asian cohorts treated with sunitinib, sorafenib, or pazopanib confirmed that IMDC retains prognostic discrimination, although absolute survival times may differ based on local practice patterns and drug availability.11, 8, 9
13. Updates and Extensions to the Original Scoring Systems
Over time, both IMDC and MSKCC models have seen refinements. IMDC has been examined in second‑line and later‑line therapy, in sarcomatoid RCC, and within IO/IO and IO/TKI trials, consistently demonstrating risk‑stratified survival despite evolving therapeutic backbones. Studies have proposed integrating additional markers—such as neutrophil‑to‑lymphocyte ratio, CRP, or genomic features—but these remain investigational and are not yet incorporated into major guidelines.12, 1, 2
14. Clinical FAQs
When should you not use the RCC Prognosis Panel?
The panel is intended for adults with advanced or metastatic RCC who are candidates for systemic therapy; it is not validated for localized, surgically cured tumors without evidence of recurrence. It should be used cautiously in patients with significant non‑cancer‑related organ dysfunction (e.g., acute inflammatory states, severe cytopenias unrelated to RCC), where laboratory derangements may not reflect tumor biology and could misclassify risk.1, 2
How does IMDC differ from MSKCC in guiding IO/TKI therapy?
IMDC was developed and validated in the targeted‑therapy era and has been incorporated into modern IO/IO and IO/TKI trials, making it the primary model embedded in current NCCN and European guidelines. MSKCC was derived from cytokine‑era populations, with different baseline treatments and survival expectations, so it is now mainly used for historical comparisons and to interpret older studies rather than to drive first‑line IO/TKI choices.4, 6, 7, 5, 2, 1
Is the RCC Prognosis Panel applicable to Asian patients?
Yes. Multiple analyses have shown that IMDC risk stratification retains prognostic value in Asian cohorts treated with VEGFR‑TKIs and other systemic therapies, with hazard ratios similar to Western populations. Nevertheless, clinicians should consider regional differences in available agents, comorbidities, and supportive care when translating median OS figures from international datasets to local practice.8, 9, 11
What is the most common mistake when calculating IMDC or MSKCC risk?
A frequent error is misclassifying patients due to inconsistent lab reference ranges—using fixed cut‑offs instead of each laboratory’s lower and upper limits of normal for hemoglobin, calcium, neutrophils, platelets, and LDH. Another common issue is failing to account for the time from diagnosis to systemic therapy, particularly in patients who had prolonged surveillance or multiple surgeries before starting systemic treatment; the RCC Prognosis Panel explicitly prompts for this to reduce oversight.3, 2, 1
Can the panel replace clinical judgment or guideline review?
No. The RCC Prognosis Panel is a decision‑support tool meant to standardize risk assessment, not a stand‑alone directive for treatment. Final management decisions should integrate histology, comorbidities, patient preferences, local guideline adaptations, and multidisciplinary input in addition to risk scores.7, 2
15. Call to Action and Next Steps
Nephrologists and oncology teams can access the Renal Cell Carcinoma (RCC) Prognosis Panel at https://oncotoolkit.com/calculator/panel-renal-risk and bookmark it for rapid use in clinics, consult services, and MDT meetings. We encourage you to test the calculator with several representative cases—such as a favorable‑risk patient starting IO/TKI, an intermediate‑risk patient with early recurrence, and a poor‑risk patient with heavy disease burden—to see how unified IMDC and MSKCC outputs can clarify therapeutic discussions.3
As you explore OncoToolkit, consider linking from this panel to related tools such as a dedicated IMDC score calculator, an RCC staging module, or IO/TKI toxicity risk calculators, using anchor text like “IMDC risk calculator,” “RCC staging tools,” or “IO/TKI toxicity prediction.” Together, these decision‑support resources can help reduce cognitive load, standardize care, and support both clinical practice and research in advanced renal cell carcinoma.